Haemoglobin Varieties:- In man probably there are at least two varieties of haemoglobin, the foetal haemoglobin (HbF) and the adult haemoglobin (HbA). Foetal haemoglobin differs chemically and spectroscopically from adult haemoglobin. It has a greater affinity for oxygen and releases CO2 more readily. This is due to some difference in the globin fraction. This property helps to compensate for the relative anoxia of foetal blood. At low 02 pressure, foetal haemoglobin can take up larger volumes of 02 than adult haemoglobin. Thus, to summarize the well-known varieties of normal humans which include Hb are HbAl (two α-chains and two β-chains), HbA2 (two α-chains and two δ-chains), HbF (two α-chains and two γ-chains), embryonic Hb (two zeta and two epsilon chains, HbAlC (glycosylated Hb), etc.

The Form in which Haemoglobin Exists in Cells
The actual state in which haemoglobin exists in the red cells is not yet fully understood. The quantity of haemoglobin in the red cells is too great for it to be in simple solution and it is also known that it is not present in crystalline form. From these observations, it has been suggested that haemoglobin remains in some combined form. It is believed that haemoglobin remains absorbed into the lipid material of the stroma and the envelope of red cells. In certain lower invertebrates, haemoglobin remains free in the plasma. Its inclusion is in the red.
CAUSES OF INCLUSION OF THE HAEMOGLOBIN IN RED CELLS
Though haemoglobin is a protein having a molecular weight of 68,000 yet it passes through the endothelial lining of the blood vascular system as well as through the normal glomerular membrane.
Haemoglobinuria is the condition when free haemoglobin is excreted through the urine. If Hb is free in the plasma then it is excreted through the urine. In plasma, Hb is present normally bound to protein at a concentration of about 5 mg/100 ml of whole blood.
• In plasma Hb remains normally as haemoglobinheptoglobin complex which cannot pass through the urine. Under normal conditions, about 100 to 150 mg of free Hb is released from red cells due to haemolysis. Hb can be bound as a haemoglobin-heptoglobin complex by the heptoglobin present in the plasma. lf released Hb exceeds the capacity of the heptoglobin to bind the haemoglobin, then it is excreted through the urine.
• The free Hb passes readily through the glomerulus and of which a certain amount is reabsorbed and the rest is excreted through the urine. During reabsorption of Hb through the tubular epithelium certain amount of it is converted into haemosiderin and excreted in the urine. So plasma Hb concentration is always associated with haemosiderinuria though the condition of haemoglobinuria may or may not be happened. Thus, if the Hb was not enclosed by the cell membrane of erythrocytes then this Hb would have been passed quickly through the glomerular membrane and excreted through the urine. Because RBC cannot pass through the glomerular membrane in normal conditions. In transport of CO2 from the tissues to the lungs the erythrocyte with its haemoglobin plays an important part, because the enzyme carbonic anhydrase is present within the erythrocyte. If haemoglobin instead of staying within the corpuscles, remains free in the plasma (as occurs in haemolysis); many injurious effects will be produced. The degree of deleterious effects will depend upon the number of red cells haemolysed and the rapidity of haemolysis. Briefly, the following ill effects will be produced:-
Haemoglobin Varieties-
1. Viscosity of blood will rise.
2. The colloidal osmotic pressure, normally about 30, will rise to 100 mm of Hg or more. This will seriously disturb the interchange of various substances in the capillary area and will also disturb the formation of urine.
3. Loss of haemoglobin will reduce the amount of blood buffers and will cause acidosis. This is all the more enhanced by the disintegration of red cells, the loss of available surface area of the erythrocytes, which plays a considerable role in maintaining the acid-base balance and the ion balance in blood.
4. Loss of haemoglobin will reduce the oxygen-carrying capacity of blood, thus producing anoxia and acidosis.
5. Bile pigments will be produced in larger amounts by the RE cells from the released haemoglobin and in this way additional pressure will be put upon the liver to deal with them.
6. While being excreted through the kidneys, haemoglobin will be precipitated in the acid urine and in this way, the kidney tubules will be blocked. This will cause serious disturbance of kidney function. As a delayed result, hypochromic anaemia will be produced.

SYNTHESIS OF HAEMOGLOBIN
Haemoglobin is synthesised inside the red cells in the bone marrow. A number of factors is necessary for the synthesis of haemoglobin. They are as follows:
1. First-class proteins ( or proteins of high biological value): It is necessary for the synthesis of the globin part of haemoglobin. Certain individual amino acids, such as histidine, phenylalanine, leucine, etc. have been found to possess a special stimulating action on haemoglobin formation. A diet containing kidney, spleen, heart and certain fruits are very helpful. Four kinds of globin peptide chains-α, β, γ and δ have been isolated. Human haemoglobin contains two α and two β-chains. The haem portions are attached to the α- and β-chains.
2. Metals
- Iron: It is an essential constituent of haemoglobin. A daily intake of 12 mg is adequate.
- Copper, manganese and cobalt: These metals, particularly copper, help in the incorporation of iron in the protoporphyrin molecules for the formation of metalloporphyrin. The ratio between Cu:Fe in the daily diet should be 1:100. Cobalt has a definite value as a constituent of vitamin B12. They act as catalytic agents.
3. Endocrine: Of the endocrines, only thyroxine is of proven value.
4. Vitamins: Vitamins C and B12 are definitely helpful in this respect. It is also believed that folic acid, riboflavin, nicotinic acid, pantothenic acid and pyridoxine also play some parts in the formation of haemoglobin.
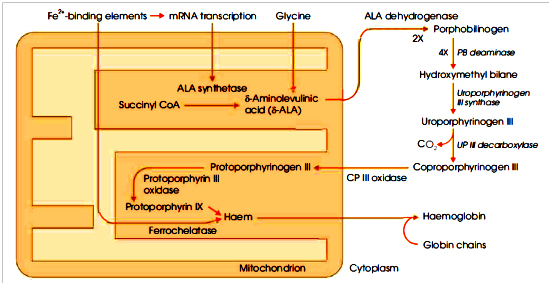
5. Porphyrins: Of the two types of porphyrins, I and III that are found in nature, the latter is utilised for haemoglobin formation. Studies with radioactive carbon show that protoporphyrin III, required for this purpose, is synthesised in the animal body from simpler substances like glycine, acetic acid, acetoacetic acid, succinic acid or any amino acid that can give rise to the formation of succinic acid during metabolism or through the tricarboxylic acid cycle.
- Glycine and succinate help in the synthesis of protoporphyrin.
- Aminolevulinic acid (ALA) is formed by the interaction of succinate and glycine. Two molecules of aminolevulinic acid after condensation form porphobilinogen (PBG).
- From porphobilinogen ultimately uroporphyrinogen III formed. On decarboxylation, the uroporphyrinogen III is converted into coproporphyrinogen III and which on oxidation ultimately gives rise to protoporphyrin III.
- Protoporphyrin III in the presence of globin and Fe++ is converted into haemoglobin. The steps involved in the synthesis of Hb (Hgb).
AMOUNT OF HAEMOGLOBIN IN NORMAL BLOOD
- The average adult figure, irrespective of sex is 14.5 g%.
- Different observers give different figures for haemoglobin.
- For instance: Sahli: 17.3%, Haldane-14.8%, Gower: 15%, and Hellige: 14% (the best method of expressing the amount of haemoglobin in blood is to mention it in the absolute figure, viz. as so many grams in 100 ml of blood.
- If it is expressed in percentage, the scale used should be mentioned).
METHODS OF ESTIMATION OF HAEMOGLOBIN
1. Tallqvist’s method: A drop of blood from the patient’s finger is soaked in a piece of filter paper and compared against a standard colour scale, before the blood on the filter paper dries up. This method does not give accurate results.
2. Haldane’s haemoglobinometer (Haldane’s modification of Gower’s method): The instrument consists of two tubes, one of which contains 20 cu mm of blood haemolysed with distilled water and saturated with CO gas. The colour of this tube is used as standard. In the other tube a little distilled water is taken and 20 cu mm of the patient’s blood, collected from the fingertip by a special pipette, is added. When blood becomes fully haemolysed, it is saturated with CO by passing coal gas through it. The colour developed is compared against that of the standard. If the colour of the unknown is stronger, it is diluted with distilled water until the tinge is the same in both.
The graduation up to which the blood has been diluted gives the percentage of haemoglobin.
3. Gower’s haemoglobinometer: Here, the standard used is a solution of picrocarmine gelatin. Otherwise, the method is the same as Haldane’s. The disadvantage of this method is that the colour of the standard gradually fades away.
4. Sahli’s method: Here, instead of distilled water (N /10) HCl is used. This converts haemoglobin into acid haematin. The colour developed is matched against that of standard and the result is obtained as in Haldane’s method.
5. Von Fleischl’s haemometer: Here, the standard used consists of a set of coloured glasses.
6. Colorimetric method: Here, the comparison is done with the help of a colorimeter.
7. For very accurate work the method of Van Slyke and Stadie should be used. In this method iron of haemoglobin is estimated and from that the corresponding amount of haemoglobin is calculated. The photoelectric methods of Haliday, Kerridge and Smith (1935) may be adopted.
8. Spectrophotometric method: It is the modern method and is based as the measurement of absorption of light at certain wavelengths of cyanmethaemoglobin formed by treating the haemoglobin with ferricyanide and then with KCN.
VARIATIONS OF HB UNDER DIFFERENT PHYSIOLOGICAL CONDITIONS
1. Age: In the foetus, the concentration is highest. At birth, the average concentration is about 23 g per 100 ml. By the end of the third month, it falls below normal, probably, because of a deficiency of iron in milk. After this gradual recovery takes place and at the end of the first year, the average amount is 12.5 g. Then it rises gradually up to normal figures.
2. Sex: In females, the amount of haemoglobin is slightly lower than in males. In adult females, the average is 13.7%, in adult males the average is 15.8%.
3. Diurnal variation: Variation of at least 10% occurs throughout the day. In the morning it is lowest; in the evening it is highest.
4. Altitude: At higher altitudes, haemoglobin percentage rises.
5. Exercise, excitement, adrenaline injection, etc. increase the amount of haemoglobin.
6. It should be noted from the above that normal variation of haemoglobin is mostly due to the alteration of a number of red cells and not due to any change in the absolute quantity of haemoglobin in each cell. Anything that alters the red cell count will alter the percentage of Hb proportionally.

Derivatives of Haemoglobin Compound
- Oxyhaemoglobin: It is a compound of haemoglobin with oxygen. Iron remains in the ferrous (Fe++) state in haemoglobin. It is not a stable compound. Oxygen may be removed when the blood is exposed to a vacuum. It has got two absorption bands between D and E.
- Methaemoglobin: It is also a compound of haemoglobin with oxygen. It can be produced after treating the blood with potassium ferricyanide. It is chocolate-brown in colour. It is a stable compound. Oxygen cannot be removed by exposing the blood to a vacuum. Iron remains in the ferric (Fe+++) state. It has got one absorption band between C and D.
- Carbohaemoglobin: It is a compound of haemoglobin with CO2. The compound is formed by the union of CO2 with the globin portion.
- Carboxyhaemoglobin or carbon monoxyhaemoglobin: Haemoglobin combined with CO instead of oxygen. It is present in blood in coal gas poisoning. It has got two absorption bands between D and E. The affinity of human haemoglobin at 38°C, for CO, is 210 times greater than O, the extremely poisonous.
- Sulphaemoglobin: It is formed by the combination of haemoglobin with H2S. The compound is very stable and is sometimes found in the blood after certain kinds of drug poisoning.
- Nitric oxide haemoglobin: Haemoglobin combined with NO instead of oxygen, is found in nitric oxide poisoning.
Derived Product
Iron Containing
- Haematin: This derivative can exist in two forms-acid and alkaline and may be prepared from haemoglobin by the action of acid or alkali. This is sometimes found in the urine in old cases of haemorrhage. It is a ferric compound.
- Acid haematin has got an absorption band between C and D. The absorption band of alkaline haematin is near D line.
- Haemin: Haemin is haematin hydrochloride. It is prepared by boiling oxyhaemoglobin with NaCl and glacial acetic acid. It is a ferric compound.
- Haemochromogen: When alkaline haematin is reduced by ammonium sulphide, this derivative is obtained. It is a ferrous compound. Haem with ferrous iron is combined with denatured globin. Of all the haemoglobin derivatives, haemochromogen possesses the most characteristic spectrum. It has a very distinct band between D and E, as well as a fainter band between E and B lines. Due to this property, this compound is often used to identify doubtful blood stains.
- Cathaemoglobin: It is a compound of haem containing ferric iron with denatured globin.
- Haem: It is a ferrous compound produced by the reduction of haematin in an alkaline solution.
Iron Containing
- Haematoporphyrin: This derivative can exist in two forms-acid and alkaline. It is prepared by mixing blood with sulphonic acid. When mixed with alkali the alkaline variety is formed. Normal urine may contain traces. It is found in the blood and urine in sulphonal poisoning and in certain cases of liver disease.
- Haemopyrrole: When haematoporphyrin is reduced, this compound is formed. It is probably a mixture of several pyrrole compounds.
- Haematodin: This compound is produced by the breakdown of haemoglobin in the body. It is found as yellowish-red crystals in the region of old blood extravasations. Some authors believe that it is identical to bilirubin.
- Bilirubin: It is the chief pigment of bile and is produced from haemoglobin in the whole of the reticuloendothelial system. From it are derived all the other bile pigments, the pigment of the stool, stercobilin; the pigments of the urine, such as urobilinogen, urobilin and urochrome.
Properties
1. Oxygen association: The most characteristic property of haemoglobin is the ease with which it combines with oxygen and dissociates from it. 100 ml of water will absorb one-third of oxygen at body temperature under atmospheric pressure. But 100 ml of blood, under the same condition, will take up 20 ml of oxygen (60 times), due to the presence of haemoglobin (about 1,200 ml of oxygen can be carried by the total amount of blood in an adult man). One gram of haemoglobin combines at normal (standard) temperature pressure (NTP) with 1.34 ml of oxygen. This corresponds to two atoms of oxygen for each atom of iron. The compound oxyhaemoglobin gives off its full oxygen content when placed in a vacuum.
2. Oxyhaemoglobin holds its oxygen loosely which can be easily displaced by many other gases forming more stable compounds, e.g. CO, NO, H2S will form carboxyhaemoglobin ( carbon on oxyhaemoglobin), nitric oxide haemoglobin, sulphaemoglobin respectively.
3. The globin part of haemoglobin directly combines with CO2 and forms carbaminocompounds.
4. Crystallisation: Haemoglobin can be easily crystallised. The form of the crystals, their solubility and the ease of crystallisation are characteristic of the species from which haemoglobin is obtained. Most blood, including human blood, form rhombic prisms or needles. Haemoglobins of different species are said to be immunologically distinct. The distinction lies in the globin part of the molecule and not in the haem part. It is known that the amino acid composition of the various globins (derived from the haemoglobin of different species) varies considerably, especially with respect to their cysteine content.
5. The haemoglobin of different species also shows a different affinity for oxygen.
6. Isoelectric pH of haemoglobin (reduced Hb) is 6.8, and that of oxyhaemoglobin is 6.6.